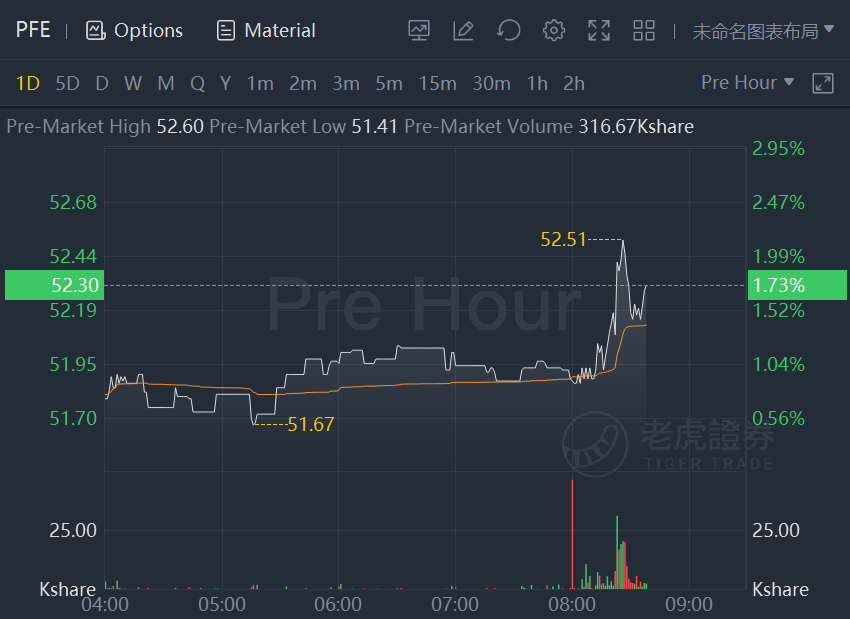
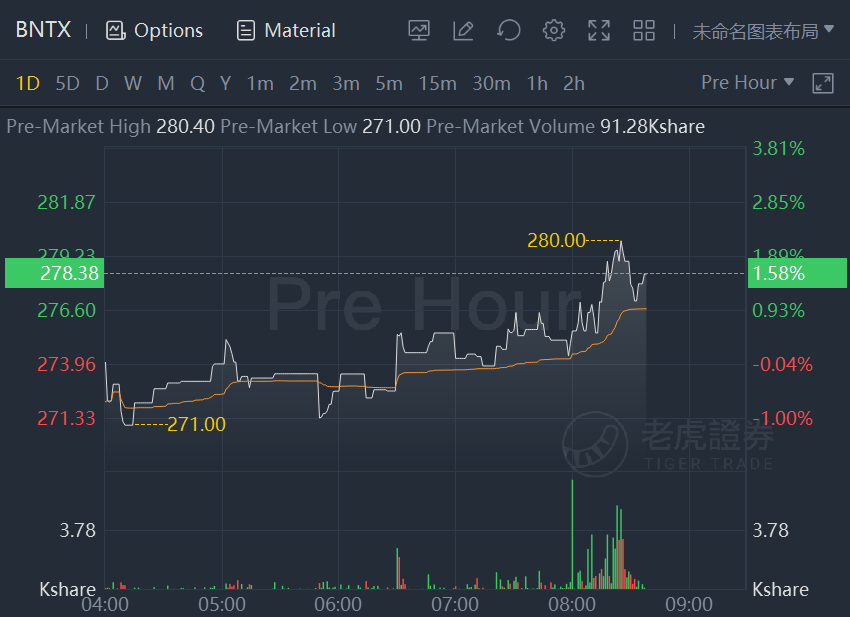
Pfizer and BioNTech SE stocks climbed about 2% in premarket trading after they received expanded U.S. FDA emergency use authorization of booster to include individuals 18 and older.
Pfizer Inc. and BioNTech SE today announced that the U.S. Food and Drug Administration (FDA) has expanded the emergency use authorization (EUA) of a booster dose of the Pfizer-BioNTech COVID-19 Vaccine to include individuals 18 years of age and older. The booster dose is to be administered at least six months after completion of the primary series, and is the same dosage strength as the doses in the primary series.
“As we near the two-year mark in our fight against COVID-19, we have reached another critical milestone with the expanded authorization of a booster dose of our COVID-19 vaccine in individuals 18 years and older,” said Albert Bourla, Chairman and Chief Executive Officer, Pfizer. “With boosters, more adults will now have the opportunity to help preserve a high-level of protection against this disease. We are grateful to the FDA for their rigorous review, and the action taken today that we hope will help accelerate our path out of this pandemic.”
“Today’s FDA decision is supported by clinical data showing robust immune responses following a booster dose of our vaccine, exceeding what has been seen even after the completion of the highly-effective two-dose primary schedule,” said Ugur Sahin, M.D., CEO and Co-founder of BioNTech. “These data suggest a booster dose of our vaccine has the potential to maintain a high-level of protection against tested variants, including Delta.”
In October, the companies announced positive topline results from the trial showing that a booster dose administered to individuals who previously received the Pfizer-BioNTech primary two-dose series demonstrated a relative vaccine efficacy of 95% when compared to those who did not receive a booster. Thus far, these are the first and only efficacy data disclosed from any randomized, controlled COVID-19 vaccine booster trial. The adverse event profile was generally consistent with other clinical safety data for the vaccine, with no new safety concerns identified.
A booster dose of the Pfizer-BioNTech COVID-19 Vaccine was previously authorized by the FDA for emergency use after completion of a primary series in individuals 65 years of age and older, individuals 18 through 64 years of age at high risk of severe COVID-19, and individuals 18 through 64 years of age with frequent institutional or occupational exposure to SARS-CoV-2, as well as eligible individuals who have completed primary vaccination with a different authorized COVID-19 vaccine.
Pfizer and BioNTech continue to supply the vaccine, including sufficient volume for boosters, under their existing supply agreement with the U.S. government, which continues through April 2022. The companies do not expect that today’s news will impact the existing supply agreements in place with governments and international health organizations around the world.